
RAMP COVID-19 antigen test gets Health Canada approval
Response Biomedical’s RAMP COVID-19 antigen test gets Health Canada approval. Response Biomedical Corp. (Response), a global provider of acute care diagnostic testing solutions, is announcing

Response Biomedical’s RAMP COVID-19 antigen test gets Health Canada approval. Response Biomedical Corp. (Response), a global provider of acute care diagnostic testing solutions, is announcing

We know much more about rapid COVID testing now than ever before. Learn three key lessons in acute care diagnostics from the pandemic.
As a medical device manufacturer, Response Biomedical (RBM) has been closely monitoring the research regarding the detection of SARS-CoV-2.
Since the onset of the pandemic, rapid testing has gone through many changes. Discover how POC testing has affected the healthcare system and what lies ahead.

Response Biomedical launches COVID-19 (SARS-CoV-2) antigen test for countries accepting CE mark, to support the growing demand for testing. Response Biomedical Corp. (Response), a global
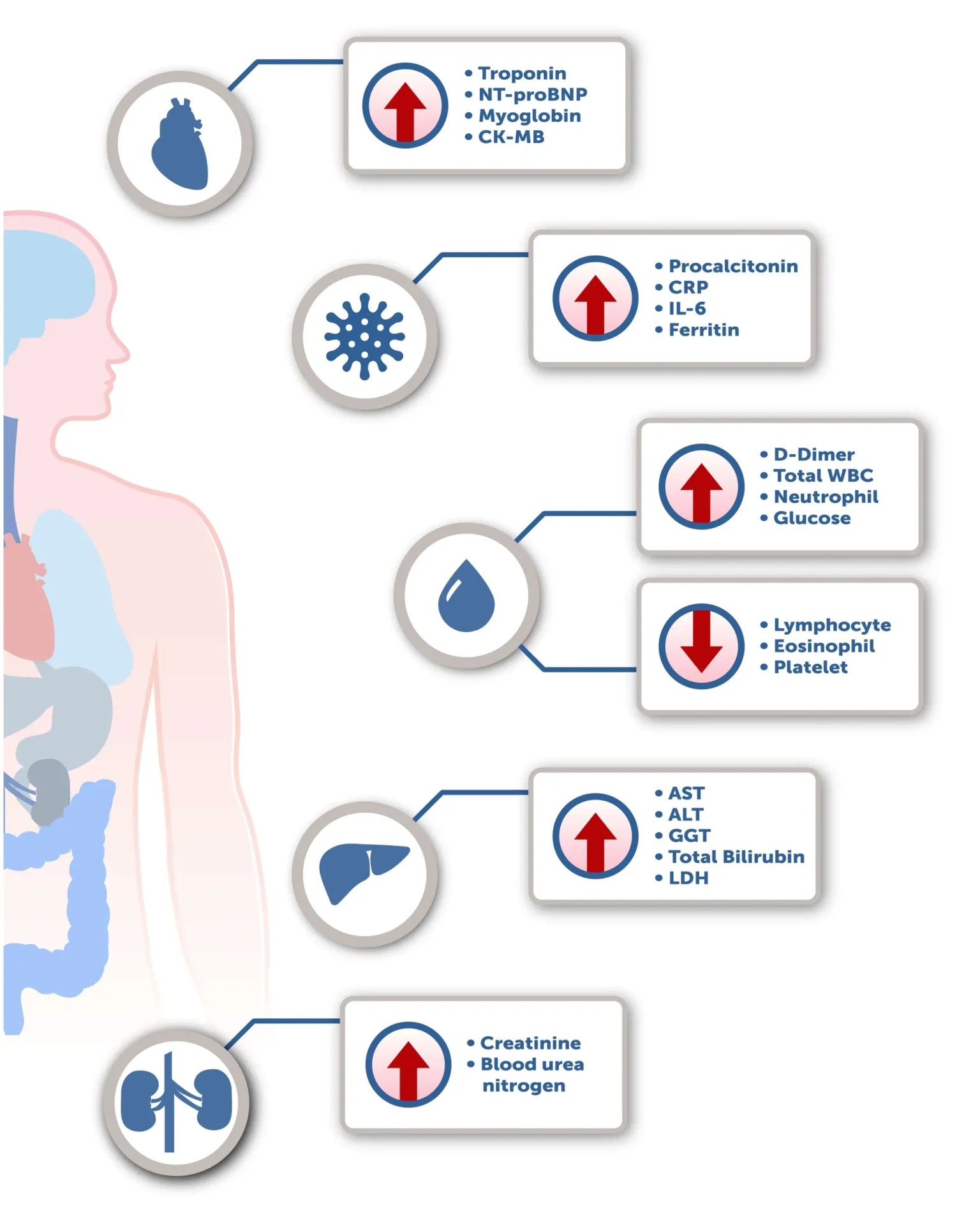
As a medical device manufacturer, we have been closely monitoring the research that is currently being conducted into the detection of SARS-CoV-2. Response Biomedical’s purpose
Response Biomedical Corp. is proud to be part of Canada’s fight against COVID-19 by developing a COVID-19 Antibody (Serology) test on the RAMP® platform. The
Response Biomedical is excited to be a part of Next Generation Manufacturing Canada’s (NGen) fight against COVID-19 by developing a 15 minute COVID-19 test on
As the amount of available information and research into COVID-19 is constantly changing, we wish to continue to provide you with updated information. Please find
Not all products are available in all regions. Contact us for availability in specific markets.
"*" indicates required fields
"*" indicates required fields