As a medical device manufacturer, we have been closely monitoring the research that is currently being conducted into the detection of SARS-CoV-2. Response Biomedical’s purpose is to improve and save as many lives as possible, and we strive to provide our channel partners with the most recent and relevant information.
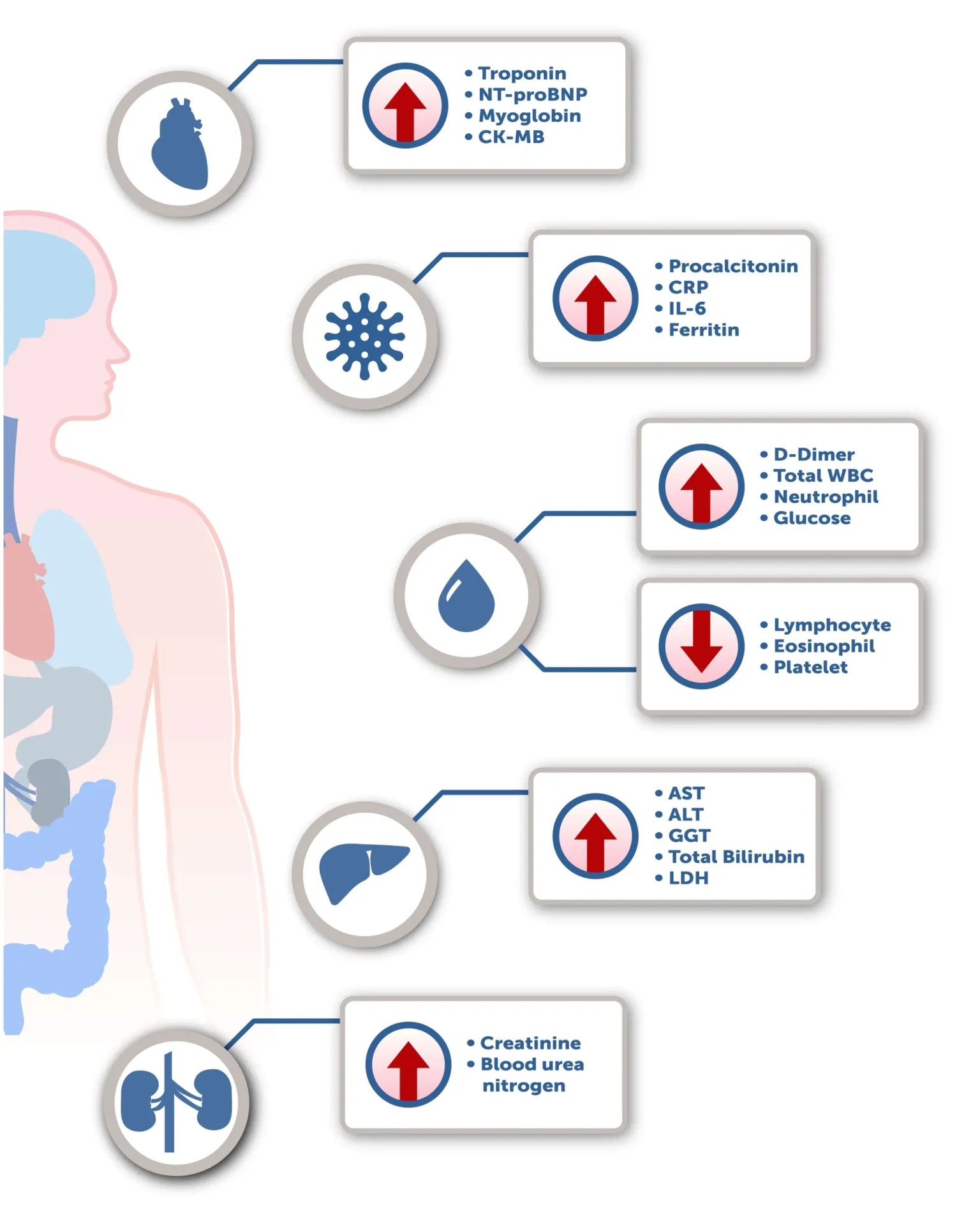
Various literature has been found which broadly defines the clinical characteristics of COVID-19. However, the laboratory abnormalities for these patients depends significantly on the patient’s clinical presentation at the time of evaluation. Most of these tests have been found useful for hospitalized patients and show a trend which indicates whether a patient is either improving or progressing to a more critical state.
As may occur in other infections, leukopenia and leukocytosis are common in COVID-19. Patients with more severe infection have had elevations of liver enzymes, muscle enzymes, C-reactive protein, lactate dehydrogenase, and ferritin. [1, 2] Several papers have also shown increased procalcitonin(PCT) and D-dimer levels in patients with a severe infection. [3,4] While not typically elevated in patients with viral infection, progressive increases in PCT values have been shown to correlate with worse prognosis, likely related to bacterial superinfection. Elevated D-dimer levels have been strongly associated with greater risk of death and thrombotic complications, most frequently deep venous thrombosis, and pulmonary embolism. [5-7] Elevated troponin levels are common in severe COVID-19 cases; while patients with comorbidities such as hypertension, CAD, COPD or diabetes are at higher risk, substantial heart damage has occurred in patients without any underlying cardiovascular disease. [8-10]
Please note, all studies mentioned in this COVID-19 update were not completed using RAMP products manufactured by Response Biomedical Corp. RAMP products have not been evaluated specifically for use with COVID-19 patients.
References:
1 Huang C; Wang Y; Li X; Ren L; Zhao J; Hu Y; Zhang L; Fan G; Xu J; Gu X; Cheng Z; Yu T; Xia J; Wei Y; Wu W; Xie X; Yin W; Li H; Liu M; Xiao Y; Gao H; Guo L; Xie J; Wang G; Jiang R; Gao Z; Jin Q; Wang J; Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020; 395(10223):497-506
https://www.thelancet.com/pdfs/journals/lancet/PIIS0140-6736(20)30183-5.pdf
2 Wu Z; McGoogan JM. Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323(13):1239–1242
https://jamanetwork.com/journals/jama/fullarticle/2762130
3 Lippi G; Plebani M. Laboratory abnormalities in patients with COVID-2019 infection. Clinical Chemistry and Laboratory Medicine (CCLM), 58(7), 1131-1134
https://www.degruyter.com/view/journals/cclm/58/7/article-p1131.xml
4 Zhang, J‐J, Dong, X, Cao, Y‐Y, et al. Clinical characteristics of 140 patients infected with SARS‐CoV‐2 in Wuhan, China. Allergy. 2020; 75: 1730– 1741.
https://onlinelibrary.wiley.com/doi/full/10.1111/all.14238
5 Bikdeli B, Madhavan M, Jimenez D, et al. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-up. J Am Coll Cardiol. 2020 Jun, 75 (23) 2950-2973.
https://www.onlinejacc.org/content/75/23/2950.full
6 Cannegieter, SC, Klok, FA. COVID‐19 associated coagulopathy and thromboembolic disease: Commentary on an interim expert guidance. Res Pract Thromb Haemost. 2020; 4: 439– 445.
https://onlinelibrary.wiley.com/doi/abs/10.1002/rth2.12350
7 Lippi G, Favaloro EJ. D-dimer is associated with severity of coronavirus disease 2019 (COVID-19): a pooled analysis. Thromb Haemost 2020; 120(05): 876-878.
https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-0040-1709650#
8 Guo T, Fan Y, Chen M, et al. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020;5(7):1-8.
https://jamanetwork.com/journals/jamacardiology/fullarticle/2763845
9 Canadian Cardiovascular Society (2020). Guidance from the CCS COVID-19 Rapid Response Team. COVID-19 and Cardiovascular Disease: What the Cardiac Healthcare Provider Should Know. https://www.ccs.ca/images/Images_2020/COVID_and_Cardiovascular_Disease_22Mar2020.pdf
10 Zheng YY, Ma YT, Zhang JY, Xie X. COVID-19 and the cardiovascular system. Nat Rev Cardiol. 2020. 17:259–260.