As a medical device manufacturer, Response Biomedical (RBM) has been closely monitoring the research regarding the detection of SARS-CoV-2. RBM’s core purpose is to improve and save lives globally and we strive to provide our channel partners with the most recent and relevant market information.
The World Health Organization (WHO) affirmed Coronavirus Disease 2019 (COVID-19) as a public health emergency of international concern and declared it as a pandemic on 11 March 2020.1
As the cases have increased so has the clinical, diagnostic, and scientific information available to help in the fight against COVID-19. One clinical finding discovered that cardiac injury is a common complication of COVID-19 and is present in up to 19.7% of patients who are hospitalized.2 Additionally, a meta-analysis of the association between NT-proBNP and mortality in patients with COVID-19 found that elevated NT-proBNP levels were associated with increased mortality in patients with COVID-19 pneumonia.3 Furthermore, the American College of Cardiology issued the following statement: “Natriuretic peptides are biomarkers of myocardial stress and are frequently elevated among patients with severe respiratory illnesses typically in the absence of elevated filling pressures or clinical heart failure. Much like troponin, elevation of BNP or NT-proBNP is associated with an unfavorable course among patients with ARDS (acute respiratory distress syndrome).”4
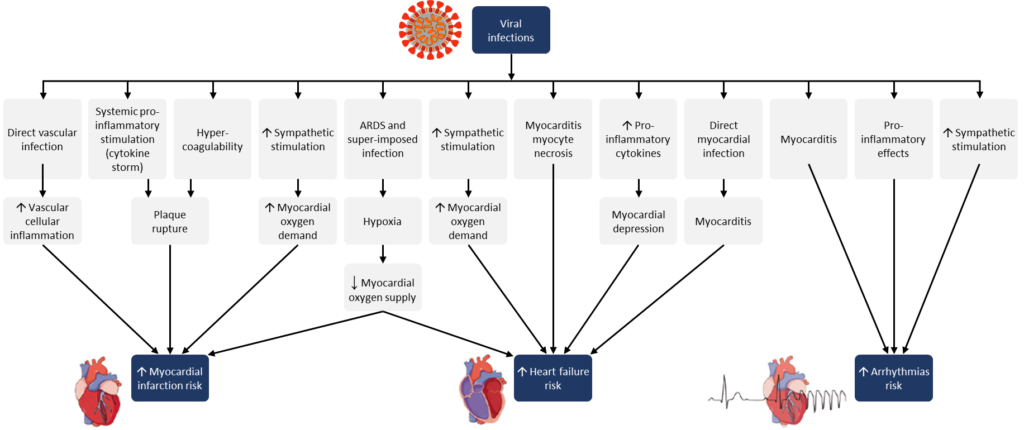
In July 2020, the American College of Cardiology issued another article regarding Heart Failure Biomarkers in COVID-19 that provides guidance on how NT-proBNP can be utilized for patients with COVID-19. The article states that COVID-19 is a systemic infection that involves multi-organ dysfunction and inflammation and consequently several biomarkers can give an abnormal result. A significant elevation in NT-proBNP could support clinical judgement regarding the involvement of the cardiovascular system in the infection and the information could be used to better triage and manage patients.5 The article also provided the figure below that shows the potential mechanisms for acute effects of viral infections on the cardiovascular system:
An additional study conducted by Dr. Juan Caro-Codón (Cardiology, Hospital Universitario La Paz, IdiPaz, CiberCV, Madrid, Spain) found that NT-proBNP is frequently elevated in patients with COVID-19. It is strongly and independently associated with mortality after adjusting for relevant confounders, including chronic HF and acute HF. Therefore, its use may improve early prognostic stratification in this condition.6
RBM recently completed a study with an outpatient cardiac clinic in Calgary, Alberta, Canada, to investigate the impact on patient, staff, and physician satisfaction levels if a patient received their NT-proBNP result within single visit. The RAMP® NT-proBNP test was used to test patient blood samples taken in the clinic and results (obtained in ~15 minutes) were shared with the patient. The study found that the use of RAMP® NT-proBNP in a physician clinic reduced patient anxiety over results, reduced the frustration of having to wait for results, improved patient understanding of their results and their condition, and increased patient’s overall satisfaction. Additionally, RAMP® NT-proBNP was found to be a user-friendly, high-quality acute care device for the management of heart failure patients. Please contact us today for a copy of the white paper (WP-CV-006).
References:
1. Domenico Cucinotta (2020) WHO Declares COVID-19 a Pandemic. Acta Biomed. 2020 Mar 19;91(1):157-160.
2. Shi S, Q. M. (2020). Association of cardiac injury with mortality in hospitalized. JAMA Cardiol, doi:10.1001.
3. Raymond Pranata, I. H. (2020). Elevated N-terminal pro-brain natriuretic peptide is associated with increased mortality in patients with COVID-19: systematic review and meta-analysis. BMJ, 387 – 391.
4. American College of Cardiology. (2020). Troponin and BNP Use in COVID-19. Washington, DC 20037: https://www.acc.org/latest-in-cardiology/articles/2020/03/18/15/25/troponin-and-bnp-use-in-covid19.
5. American College Of Cardiology. (2020). Heart Failure Biomarkers in COVID-19. Washington, DC 20037: American College of Cardiology.
6. Caro-Codón, J. (2021). Characterization of NT-proBNP in a large. European Journal of Heart Failure, 1-1.